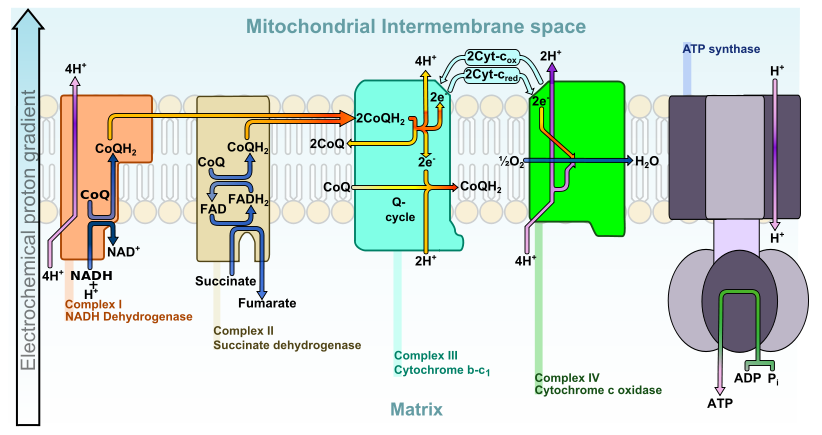
What is Cytochrome C Reductase?
Cytochrome c reductase is a large enzyme found in the power plants of the cell, the mitochondria. Approximately 78 kDaltons in size, this enzyme, also known as NADH dehydrogenase, is critical for ATP production via the electron transport chain.
Adenosine triphosphate (ATP) provides the energy necessary for the extensive and varied functions of cells.
What does Cytochrome C Reductase Do?
Cytochrome c reductase belongs to a family of enzymes called oxidoreductases which catalyze the shuttling of electrons from one molecule to another. Specifically, cytochrome c reductase functions to transfer electrons between nicotinamide adenine dinucleotide (NAD) cofactors and cytochrome C protein acceptor molecules.
Structurally, cytochrome c reductase is a flavoprotein, which means it contains a nucleic acid derivative of riboflavin as a vital component of the enzyme structure. Termed flavin dinucleotide, this region is critical for shuttling the electrons between NAD and cytochrome c and is key for forming ATP.
Cytochrome c is a small protein which contains an iron ring structure (heme) as part of the molecule. It is critically tied to complexes which carry out the electron transport chain and cellular respiration within the mitochondria.
In addition to various types of electron transport chain research studies, cytochrome c reductase has been used in manufacturing biosensors for detecting nitrite.
Cytochrome C Reductase Enzyme from Porcine Heart
For over 15 years our chief scientist, Dr. Randy Meyer, has been manufacturing and supplying quality cytochrome c reductase to companies such as Millipore Sigma. We are pleased to now also offer our cytochrome c reductase direct. Offering the same quality you’ve come to expect at attractive manufacturer-direct prices.
IntegriZyme, LLC isolates and purifies quality enzymes in the U.S. from U.S.-sourced animals.
Cytochrome c reductase is available for purchase in 10, 50, and 250 unit vials. We also have larger quantities available and can custom package in sizes to fit your needs. Please inquire.
Cytochrome C Reductase Specifications
CAS Number 9027-14-9; Enzyme Commission (EC) Number 1.6.99.3
Activity:
≥1.0 unit/ mg protein
One unit will reduce 1.0 μmole of oxidized cytochrome c per min at pH 8.5 at 25°C
Protein Content:
> 40% (Lowry)
Form:
Lyophilized powder. Crude, containing potassium phosphate, pH approximately 7
Storage:
-20C
For research, laboratory or manufacturing use only. Not for drug or household use.